GLP-2 (TZ)
Dual GIP + GLP-1 Receptor Agonist
CAS: 2023788-19-2
Formula: C225H348N48O68
M.W.: 4813.5251 g/mol
Purity: 99%+ HPLC Verified
Ref No.:G2T-0005VI
Ref No.:G2T-0010VI
For Research Use Only (RUO)
Not for human or veterinary use.

GLP-2 (TZ)
Category
GLP / Metabolic Research
CAS Number
2023788-19-2
Form Factor
Lyophilized Powder
In stock, ready to ship
Ships same day before 12pm PST
Products are shipped in lyophilized powder form.
GLP-2 (TZ) is a dual-agonist peptide activating both GIP and GLP-1 receptors. This “twincretin” mechanism achieves greater weight reduction and glycemic control than GLP-1 single-agonists, with some participants achieving weight loss exceeding 20% of body weight in clinical trials.
Description
GLP-2 (TZ) is a dual-agonist peptide activating both GIP and GLP-1 receptors. This "twincretin" mechanism achieves greater weight reduction and glycemic control than GLP-1 single-agonists, with some participants achieving weight loss exceeding 20% of body weight in clinical trials.Chemical Information
CAS Number
2023788-19-2
Purity
99%+ HPLC Verified
Molecular Formula
C225H348N48O68
Molar Mass
4813.5251 g/mol
Form Factor
Lyophilized Powder
Synonyms
Research Applications
- Metabolic Research: GLP-2 (TZ) is the leading dual GIP/GLP-1 receptor agonist studied in metabolic research. Its simultaneous activation of both incretin receptors produces superior weight reduction and glycemic control compared to single-agonist peptides. Research examines the molecular basis for this synergy, including differential receptor trafficking, biased agonism, and tissue-specific signaling outcomes.
- Endocrinology: In endocrine research, GLP-2 (TZ) is studied for its effects on insulin sensitivity, beta-cell function, and glucagon suppression. The dual-agonist mechanism produces more robust glucose-dependent insulin secretion than GLP-1 agonism alone, with the GIP component potentially contributing to improved lipid metabolism and adipose tissue remodeling. Studies examine dose-response relationships and long-term metabolic adaptation.
- Hepatology: GLP-2 (TZ) is actively studied for non-alcoholic fatty liver disease (NAFLD) and steatosis research. Clinical data demonstrate significant reductions in liver fat content and improvement in hepatic inflammatory markers. Research investigates whether the dual-agonist mechanism provides hepatoprotective effects beyond those attributable to weight loss alone, including direct effects on hepatic lipid metabolism and insulin signaling.
- Cardiology: Cardiovascular outcome research with GLP-2 (TZ) examines its effects on atherosclerotic cardiovascular disease risk factors. Studies show improvements in blood pressure, lipid profiles (triglycerides, LDL-cholesterol), and inflammatory biomarkers. Ongoing large-scale cardiovascular outcomes trials are evaluating whether these metabolic improvements translate to reduced rates of major cardiovascular events.
Laboratory Preparation Protocol
- 1 Remove the flip-off cap and wipe the rubber stopper with an alcohol swab.
- 2 Using a sterile syringe, draw the desired volume of bacteriostatic water (BAC water).
- 3 Insert the needle into the vial at an angle and slowly dispense the water down the inside wall — do not spray directly onto the powder.
- 4 Gently swirl (do not shake) the vial until the powder is fully dissolved and the solution appears clear, as shown in the vial image.
- 5 Label the vial with the reconstitution date and concentration.
Storage Conditions
Unreconstituted
Store at -20°C to 4°C (freezer or refrigerator). Stable for 24+ months.
Reconstituted
Refrigerate at 2–8°C. Use within 30 days for optimal stability.
Avoid
Repeated freeze-thaw cycles, direct sunlight, and temperatures above 25°C.
Always note the reconstitution date on the vial label to track shelf life accurately.
Safety & Compliance
- Wear nitrile gloves and safety goggles when handling research compounds.
- Work in a clean, controlled environment (laminar flow hood recommended).
- Dispose of sharps and used materials in approved biohazard containers.
- This product is for laboratory research use only (RUO) — not for human or veterinary use.
Quality Assurance Standards
Each production lot is subjected to rigorous analytical evaluation to meet pharmaceutical-grade specifications. Full testing results and procedural documentation are furnished through the accompanying Certificate of Analysis (COA) with every shipment.
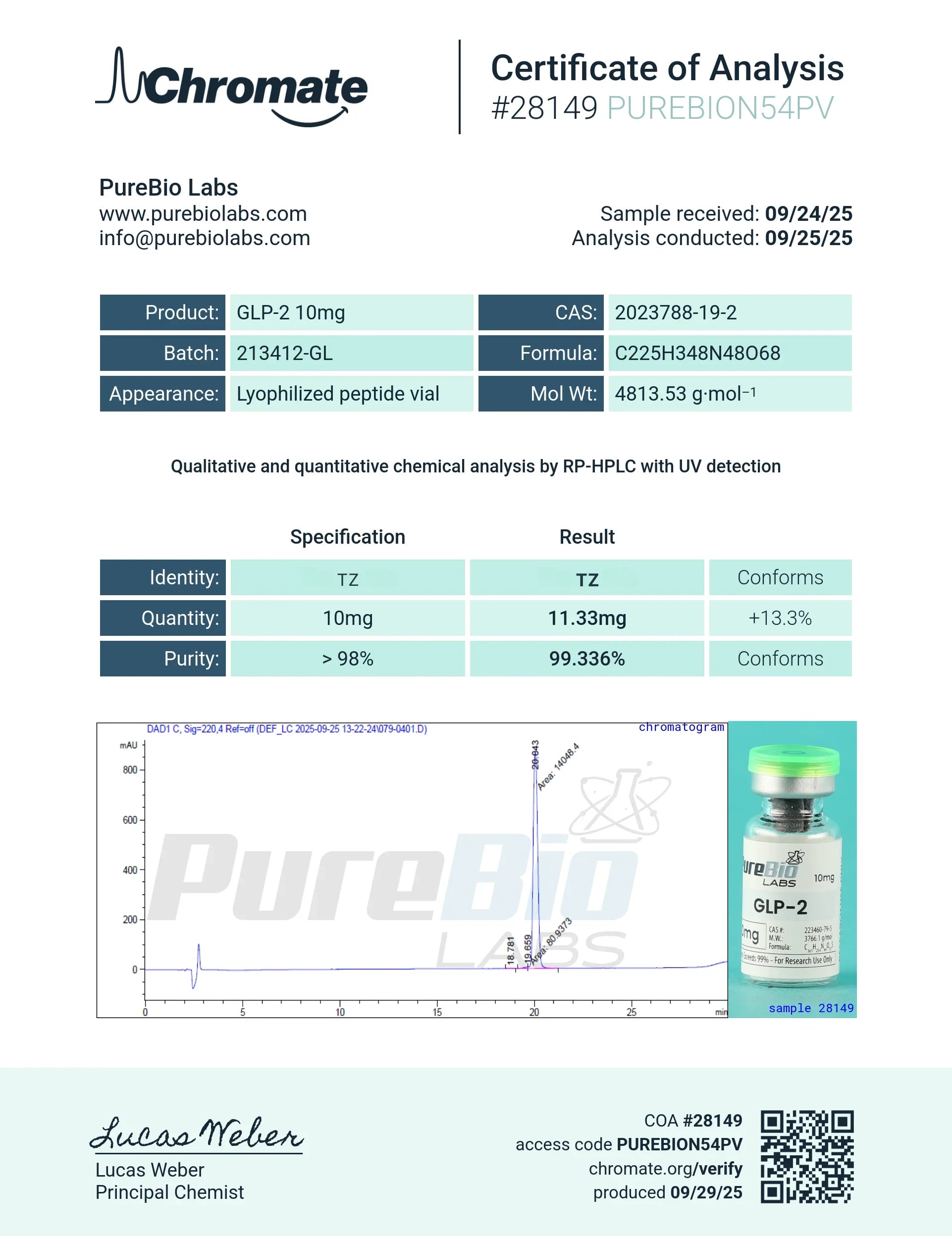
Certificate of Analysis
Latest COA for GLP-2 (TZ) — independently tested via HPLC and Mass Spectrometry.
Testing Methodology
Every batch undergoes a multi-stage analytical pipeline before release. Results are compiled into the Certificate of Analysis (COA) included with each order.
- • HPLC — High-Performance Liquid Chromatography confirms peptide purity ≥98%.
- • Mass Spectrometry — Validates molecular identity and correct sequence.
- • Endotoxin (LAL) — Limulus Amebocyte Lysate assay ensures endotoxin levels are within safe limits.
- • Sterility — Microbial contamination testing per USP standards.
Learn More About Peptides
Explore our research library covering purity, synthesis, storage, solubility, and more.
Research Database
Browse peer-reviewed studies, clinical trials, and scientific sources behind our peptide resources.
You May Also Like
Stacking Considerations
GLP-2 (TZ) targets intestinal mucosal growth, and pairs with GI-protective and metabolic peptides.
-

 $59.99 – $79.99
$59.99 – $79.99
BPC-157
Tissue Repair / Regenerative Research View DetailsThis product has multiple variants. The options may be chosen on the product page -

 $74.99
$74.99
LL-37
GH / Neuro / Misc Research View DetailsThis product has multiple variants. The options may be chosen on the product page -

 $124.99
$124.99
Semax (Spray)
GH / Neuro / Misc Research View DetailsThis product has multiple variants. The options may be chosen on the product page -

 $99.99
$99.99
NAD+ (Spray)
GH / Neuro / Misc Research View DetailsThis product has multiple variants. The options may be chosen on the product page
Complementary GI Healing
Antimicrobial gut immune support
Popular Product
Popular Product
Description
GLP-2 (TZ) is a dual-agonist peptide activating both GIP and GLP-1 receptors. This "twincretin" mechanism achieves greater weight reduction and glycemic control than GLP-1 single-agonists, with some participants achieving weight loss exceeding 20% of body weight in clinical trials.Chemical Information
CAS Number
2023788-19-2
Purity
99%+ HPLC Verified
Molecular Formula
C225H348N48O68
Molar Mass
4813.5251 g/mol
Form Factor
Lyophilized Powder
Synonyms
Research Applications
- Metabolic Research: GLP-2 (TZ) is the leading dual GIP/GLP-1 receptor agonist studied in metabolic research. Its simultaneous activation of both incretin receptors produces superior weight reduction and glycemic control compared to single-agonist peptides. Research examines the molecular basis for this synergy, including differential receptor trafficking, biased agonism, and tissue-specific signaling outcomes.
- Endocrinology: In endocrine research, GLP-2 (TZ) is studied for its effects on insulin sensitivity, beta-cell function, and glucagon suppression. The dual-agonist mechanism produces more robust glucose-dependent insulin secretion than GLP-1 agonism alone, with the GIP component potentially contributing to improved lipid metabolism and adipose tissue remodeling. Studies examine dose-response relationships and long-term metabolic adaptation.
- Hepatology: GLP-2 (TZ) is actively studied for non-alcoholic fatty liver disease (NAFLD) and steatosis research. Clinical data demonstrate significant reductions in liver fat content and improvement in hepatic inflammatory markers. Research investigates whether the dual-agonist mechanism provides hepatoprotective effects beyond those attributable to weight loss alone, including direct effects on hepatic lipid metabolism and insulin signaling.
- Cardiology: Cardiovascular outcome research with GLP-2 (TZ) examines its effects on atherosclerotic cardiovascular disease risk factors. Studies show improvements in blood pressure, lipid profiles (triglycerides, LDL-cholesterol), and inflammatory biomarkers. Ongoing large-scale cardiovascular outcomes trials are evaluating whether these metabolic improvements translate to reduced rates of major cardiovascular events.
Laboratory Preparation Protocol
- 1 Remove the flip-off cap and wipe the rubber stopper with an alcohol swab.
- 2 Using a sterile syringe, draw the desired volume of bacteriostatic water (BAC water).
- 3 Insert the needle into the vial at an angle and slowly dispense the water down the inside wall — do not spray directly onto the powder.
- 4 Gently swirl (do not shake) the vial until the powder is fully dissolved and the solution appears clear, as shown in the vial image.
- 5 Label the vial with the reconstitution date and concentration.
Storage Conditions
Unreconstituted
Store at -20°C to 4°C (freezer or refrigerator). Stable for 24+ months.
Reconstituted
Refrigerate at 2–8°C. Use within 30 days for optimal stability.
Avoid
Repeated freeze-thaw cycles, direct sunlight, and temperatures above 25°C.
Always note the reconstitution date on the vial label to track shelf life accurately.
Safety & Compliance
- Wear nitrile gloves and safety goggles when handling research compounds.
- Work in a clean, controlled environment (laminar flow hood recommended).
- Dispose of sharps and used materials in approved biohazard containers.
- This product is for laboratory research use only (RUO) — not for human or veterinary use.
Quality Assurance Standards
Each production lot is subjected to rigorous analytical evaluation to meet pharmaceutical-grade specifications. Full testing results and procedural documentation are furnished through the accompanying Certificate of Analysis (COA) with every shipment.
Certificate of Analysis
Latest COA for GLP-2 (TZ) — independently tested via HPLC and Mass Spectrometry.
Testing Methodology
Every batch undergoes a multi-stage analytical pipeline before release. Results are compiled into the Certificate of Analysis (COA) included with each order.
- • HPLC — High-Performance Liquid Chromatography confirms peptide purity ≥98%.
- • Mass Spectrometry — Validates molecular identity and correct sequence.
- • Endotoxin (LAL) — Limulus Amebocyte Lysate assay ensures endotoxin levels are within safe limits.
- • Sterility — Microbial contamination testing per USP standards.
Learn More About Peptides
Explore our research library covering purity, synthesis, storage, solubility, and more.
Research Database
Browse peer-reviewed studies, clinical trials, and scientific sources behind our peptide resources.